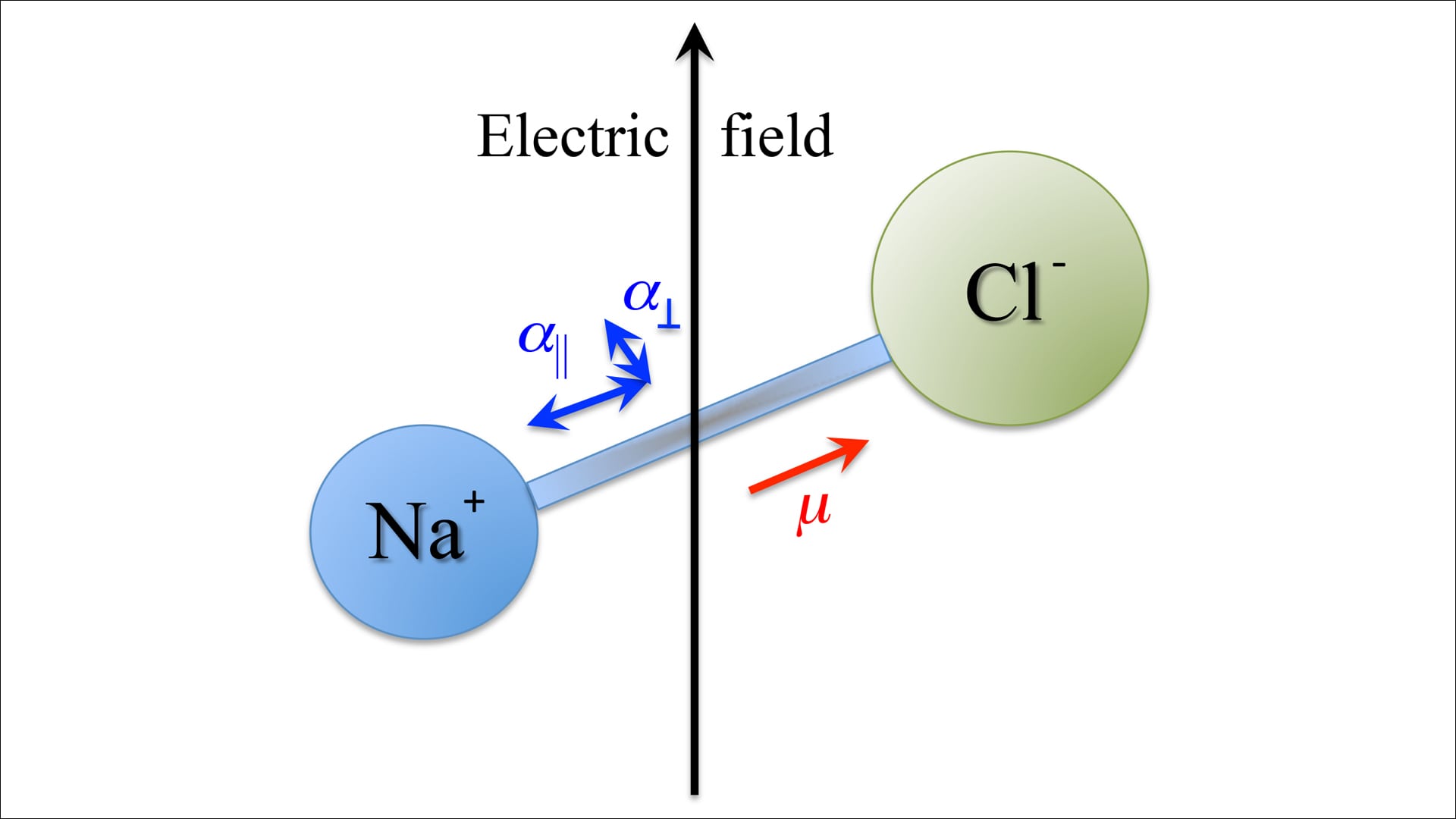
When you put a substance that does not have an electric charge in a uniform electric field it starts to get polarized. This happens because the charges inside its molecules get a bit mixed up. Usually without any field these molecules do not have a dipole moment because their positive and negative charges are centered in the same spot.
When an electric field is applied the negatively charged electrons move a bit in the opposite direction of the field while the positively charged nucleus moves in the direction of the field. This small movement creates a temporary dipole moment in the molecule.
For electric fields this temporary dipole moment is directly related to the strength of the applied field and can be written as
p = \alpha E
where \alpha is a measure of how easily the molecule can be polarized.
In a sample many molecules go through this effect leading to an overall polarization. This is described by the polarization vector P, which’s the dipole moment per unit volume. How much polarization happens depends on how strong the electric field’s how easily the molecules can be polarized.
The electric field is the same so the forces on the positive and negative charges cancel each other out. As a result there is no force on the molecule and it does not move as a whole. Only the charges, inside the molecule get rearranged.
So in an electric field a non-polar substance temporarily gets dipole moments and becomes polarized but it does not experience any overall motion because of the electric field.